|
Typically, the anomeric carbon undergoes mutarotation in solution, and the result is an equilibrium mixture of α and β configurations. In the hemiketal formation, C6-OH (or C5 - OH) of the fructose combines (through nuclcophilic addition reaction with C2-Keto group. 
Since fructose contains a keto group, it forms an intramolecular hemiketal. The anomeric carbon atoms 2 and 1 are chiral, and two configurations are possible for the OH group. Hemiacetal formation between carbons 6 and 1 leads to the pyranose structure. It is the opposite in an l-configuration furanose. Correct Answer - B C2 is the anomeric carbon in frustose. Cyclic hemiacetal (hemiketal) formation between carbon 5 and carbon 2 of fructose leads to the furanose structure. About Transcript Explore how chains of carbon atoms form carbohydrates, discover the magic of intramolecular reactions, and learn about the formation of stable rings. In a d-configuration furanose, alpha configuration has the hydroxy pointing down, and beta has the hydroxy pointing up. The furanose ring will have either alpha or beta configuration, depending on which direction the anomeric hydroxy group is pointing. In an l-configuration furanose, the substituent on the highest numbered chiral carbon is pointed downwards out of the plane, and in a D-configuration furanose, the highest numbered chiral carbon is facing upwards. The highest numbered chiral carbon (typically to the left of the oxygen in a Haworth projection) determines whether or not the structure has a d-configuration or L-configuration. The furanose ring is a cyclic hemiacetal of an aldopentose or a cyclic hemiketal of a ketohexose.Ī furanose ring structure consists of four carbon and one oxygen atom with the anomeric carbon to the right of the oxygen. The wavy bond indicates a mixture of β-ribofuranose and α-ribofuranose. Structural properties The chemical structure of ribose in its furanose form. The name derives from its similarity to the oxygen heterocycle furan, but the furanose ring does not have double bonds. Recent developments in (13) C NMR spectrometry have allowed the determination of intramolecular (13) C/(12) C ratios with high precision. The position on the anomeric carbon is the more stable. 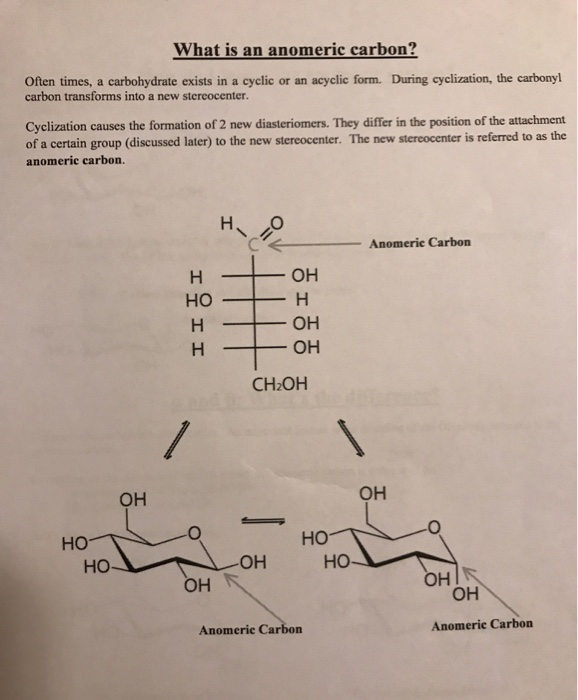
Figure 6: Sucrose has no available anomeric carbon. 
Only carbon not already an alcohol is the anomeric carbon (C1/C2). Well, sucrose cant freely equilibriate as its 2 anomeric carbons are already involved in the glycosidic linkage. Fructose (Fru) is the ketose form of glucose. A furanose is a collective term for carbohydrates that have a chemical structure that includes a five-membered ring system consisting of four carbon atoms and one oxygen atom. Galactose forms a (furanose/pyranose) ring structure whereas fructose forms. If the anomeric carbon is not involved in any kind of bond, it is free to equilibriate to its straight chain or 2 cyclic isomeric forms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
